|
1/14/2024 0 Comments Keynote 189 clinical trialAll patients were required to provide a tumor sample for determination of programmed death ligand 1 (PD-L1) status and could not have previously received systemic therapy for advanced disease. Approval based on results from the Phase 3 KEYNOTE-966 trial, which demonstrated significant overall survival benefit in these patients versus chemotherapy alone Approval marks sixth gastrointestinal cancer indication for KEYTRUDA-based regimens in the US Merck (NYSE: MRK), known as MSD outside of the United States and Canada, today announced that the U.S. Here we review phase 3 clinical trials utilizing immunotherapy in the first line for treatment of NSCLC, including Pembrolizumab in KEYNOTE-024, KEYNOTE-042, KEYNOTE-189 and KEYNOTE-407 Nivolumab and Ipilimumab in CHECKMATE-227 and CHECKMATE 9LA and Atezolizumab in IMpower110, IMpower130 and IMpower150.ĬTLA-4 chemotherapy immune checkpoints inhibitors lung cancer monoclonal recombinant antibodies programmed death receptor. Patients in KEYNOTE-189 and KEYNOTE-407 had stage IV (i.e., metastatic) disease patients in KEYNOTE-021 cohort G had stage IIIB or IV disease. Patients with PD-L1 1% immunotherapy doublet, Nivolumab and Ipilimumab, or single agent immunotherapy combined with chemotherapy. 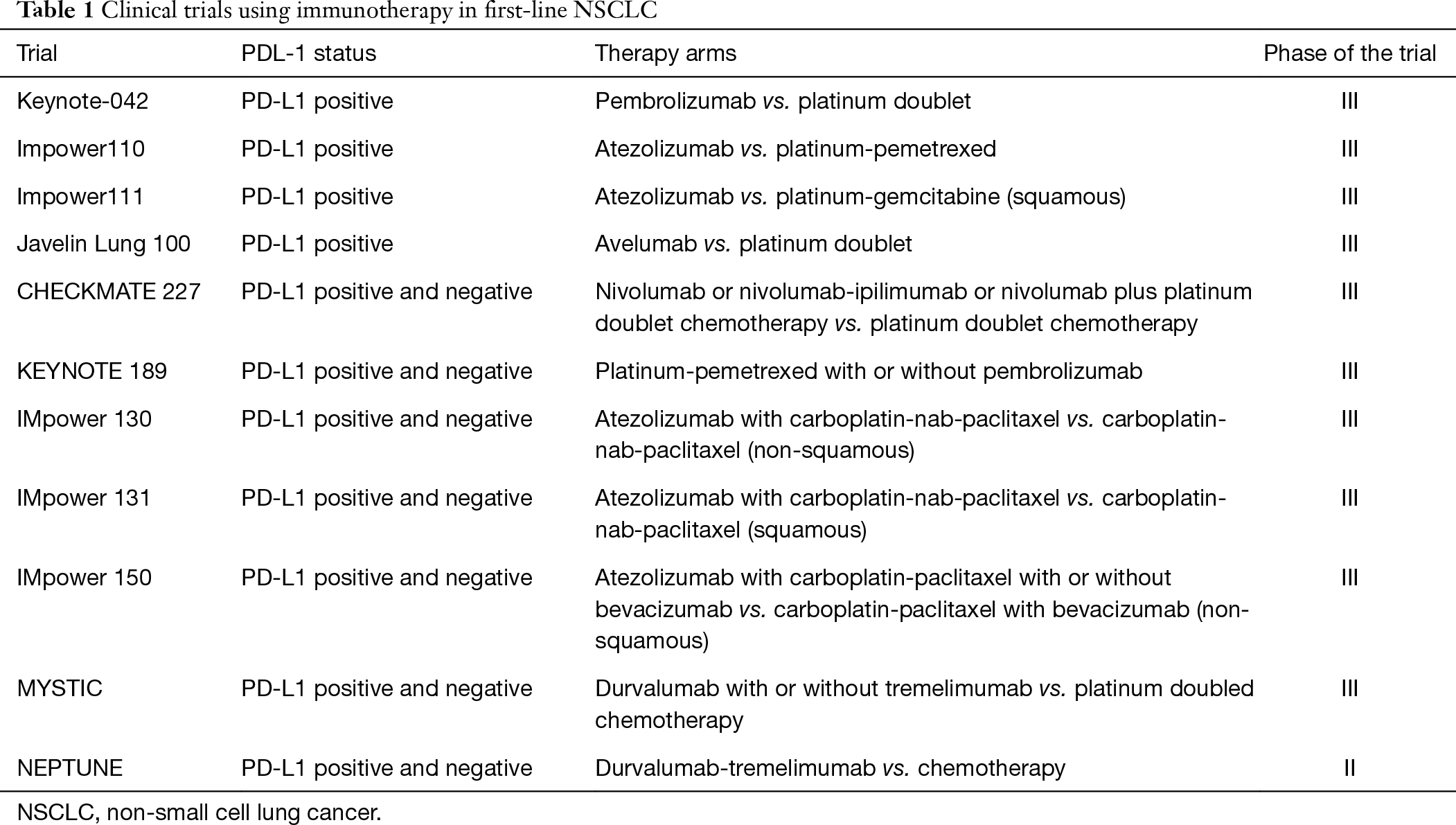 
109 A similar phenomenon also occurs in other tumor types such as. 107, 108 However, other studies have put forward different opinions that NSCLC patients with low or no expression of PD-L1 can also benefit from anti-PD-1 treatment. Patients with tumor expression levels of PD-L1 ≥ 50% are candidates for treatment with single agent Pembrolizumab or Atezolizumab. The results of KEYNOTE-189 and KEYNOTE-407 showed that OS has significant benefits regardless of PD-L1 expression. The inclusion criteria differ between the various clinical trials, including the cut-off levels of PD-L1 expression on tumor cells, and the tumor histology (squamous or non-squamous). Multiple phase 3 studies have tested different medications targeting programmed death receptor 1 (PD-1), programmed death-ligand 1 (PD-L1), cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), with or without chemotherapy. 
Immunotherapy for non-small cell lung cancer (NSCLC) is incorporated increasingly in first line treatments protocols.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
